Filenews 8 May 2025
Cyprus' position in patient access to innovative medicines remains low among European countries, according to the findings of the European W.A.I.T. (Waiting to Access Innovative Therapies) Indicator[1] of the European Federation of Pharmaceutical Industries and Associations (EFPIA) for the period 2020-2023.
Specifically, as reported by the Cyprus Association of Pharmaceutical Research and Development Companies KEFEA, our country ranks 22nd among 36 countries of the EU and the European Economic Area, a fact that highlights the timeless challenges in the approval and marketing of new pharmaceutical therapies in the Cypriot market.
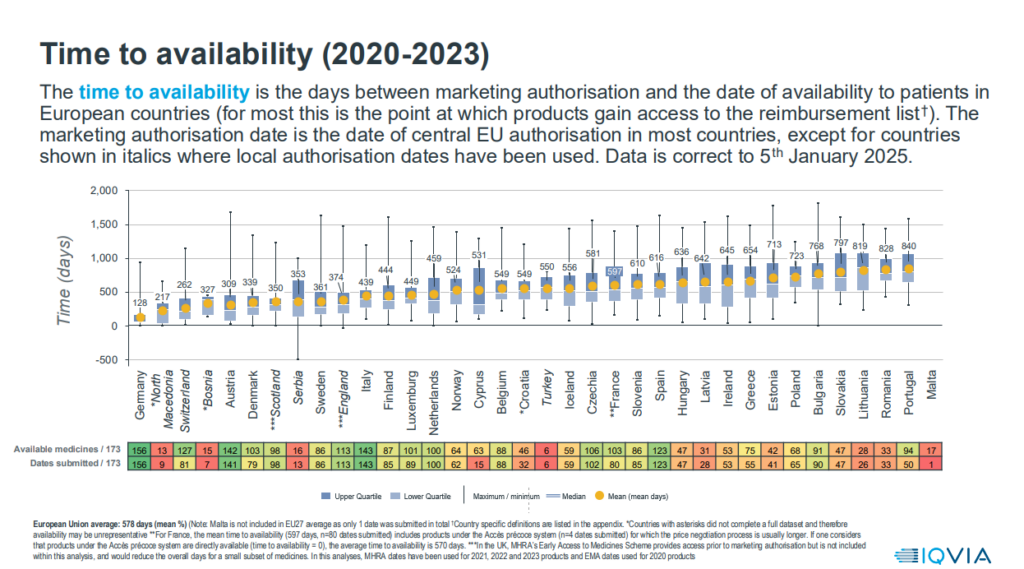
Based on the data from the survey, the results of which were published on 7 May 2025, there are still large differences in the time patients have access to new medicines (from 128 days in Germany to 840 days in Portugal with an average of 518 days). In Cyprus, the average time from approval to availability of innovative medicines reaches 531 days, while for some treatments the time has reached up to 1,300 days.
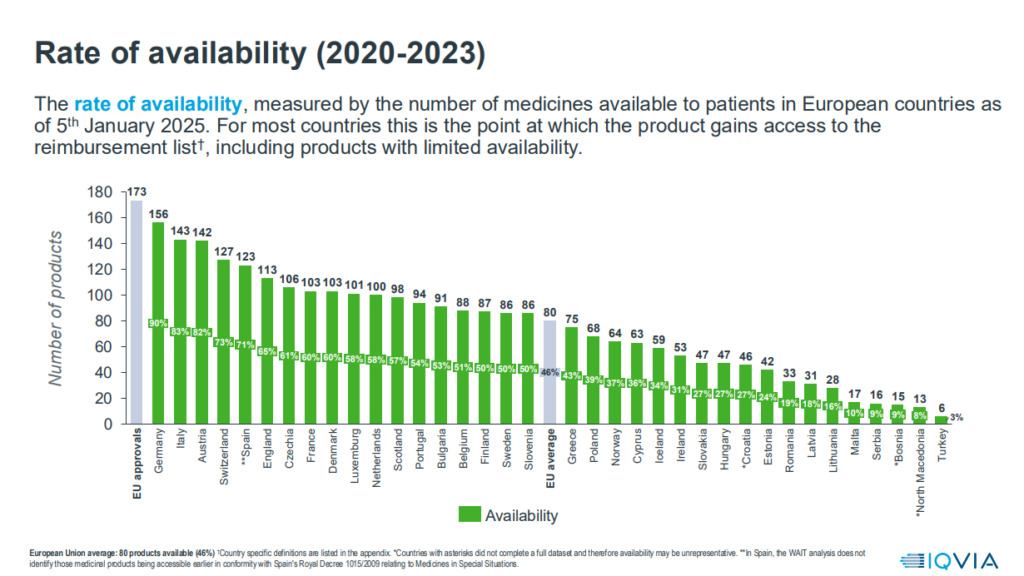
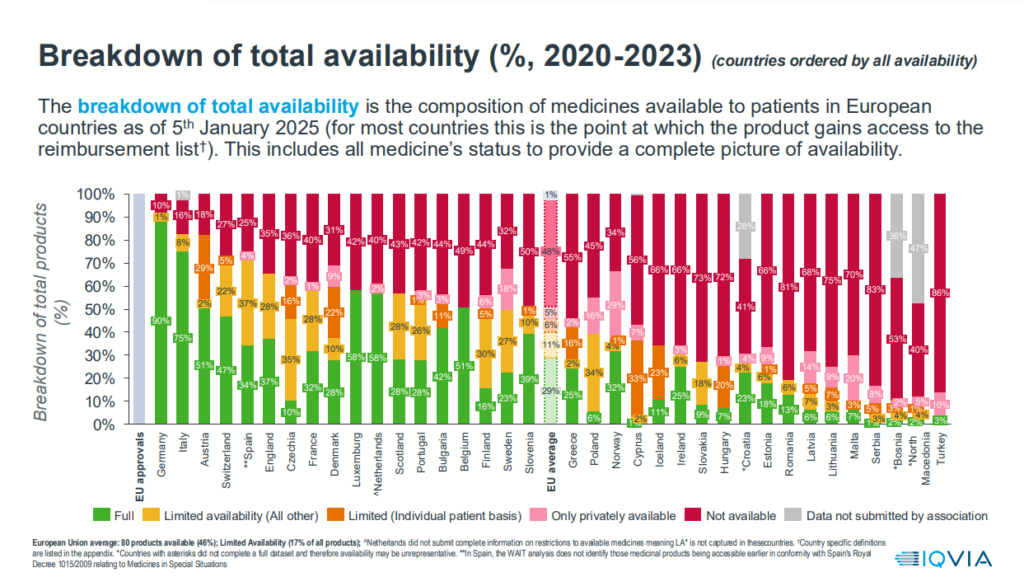
As far as the availability of medicines is concerned, only 63 innovative medicines are available in Cyprus out of a total of 173 (36%) that have secured approval from the competent European Authorities during this period. The average among the 36 countries is 46% (80 innovative medicines), while Germany still holds the lead, providing access to 156 of the 173 approved innovative medicines.
It is noted that access to innovative medicines and treatments – compared to 99% in Germany – while for 90% of formulations there is limited access or a specific procedure is provided for each occasion, through the Nominal Claims Committee (EOA).
Furthermore, the most specialised medicines for cancer or so-called orphan medicines, which usually concern rare diseases, are available to patients who need them only through limited access and mainly through the EDA. None of them, according to the survey, have been fully integrated into the compensation system.
As KEFEA reports, it has repeatedly pointed out the problems of access and availability of innovative medicines and treatments in our country, while proposing feasible and immediate solutions that facilitate patients' access to all medicines.
Six years after the introduction of the GHS, the problems with the availability of innovative medicines have not been resolved. The decision of the Ministry of Health to transfer the EDA under the Health Insurance Organization (HIO) has not only intensified delays in the provision of medication to patients who need it immediately, but has also caused delays in the completion of pending evaluations. essentially further inflating an already important problem.
"As KEFEA, we express our particular concern about the serious possibility that a gap will arise in the availability of new, innovative and not only, treatments in our country, if the situation does not improve immediately".
At the same time, they call on the competent authorities, both in the HIO and in the Ministry of Health, to take decisions and proceed with actions, the immediate implementation of which will normalize the situation with the already accumulated volume of medicines to be evaluated, in order to give the opportunity to evaluate the new applications and to comply with the timetables provided for by the legislation.